 
Electrochemical mass spectrometry measurements of CO reduction on two Cu facets led Schouten et al. observed with in situ Fourier transform infrared spectroscopy a hydrogenated dimer intermediate, *CO–*COH (asterisk means adsorbed, and the element symbol next to the asterisk is the type of atom directly bound to the surface), on Cu(100) but not on Cu(111) ( 12). To discover and design better CO 2RR electrocatalysts, many experimentalists have tried to detect reaction intermediates and to propose reaction mechanisms leading to C–C coupling on Cu. However, Cu requires high overpotentials to achieve reasonable reaction rates, and it exhibits poor selectivity toward the above-mentioned C 2 products, limiting its practical applications ( 3, 7, 10, 11). 
The major C 2 products of Cu-catalyzed CO 2R are ethylene and ethanol, along with other minor products (glycolaldehyde, acetic acid, acetaldehyde, and ethylene glycol) ( 10). This work demonstrates the importance of applying qualitatively and quantitatively accurate quantum mechanical method to simulate electrochemistry in order ultimately to shed light on ways to enhance selectivity toward C 2+ product formation via CO 2RR electrocatalysts. In contrast, standard DFT approximations arrive at qualitatively different conclusions, namely, that only *CO and *COH will prevail on the surface and their C–C coupling paths produce only *COH–*COH and *CO–*CO, with a preference for the first product. The most kinetically favorable path forms *COH–CHO. The three kinetically feasible pathways involving these species yield all three possible products: *COH–CHO, *COH–*COH, and *OCH–*OCH. We predict that two adsorbed hydrogenated CO species, either *COH or *CHO, are necessary precursors for C–C bond formation. Here, we apply embedded correlated wavefunction (ECW) theory, which regionally corrects the electron exchange-correlation error in density functional theory (DFT) approximations, to examine multiple C–C coupling steps involving adsorbed CO (*CO) and its hydrogenated derivatives on the most ubiquitous facet, Cu(111). Foundational to the rational design of efficient, selective CO 2RR electrocatalysts is mechanistic analysis of the best metal catalyst thus far identified, namely, copper (Cu), via quantum mechanical computations to complement experiments. Oxygen is a nonpolar diatomic molecule with a 180-degree bond angle.īoth oxygen atoms in the molecule have the same electronegativity value, and both atoms share identical ratios of bound shared electrons, making the O2 molecule nonpolar in nature.The electrochemical CO 2 reduction reaction (CO 2RR) powered by excess zero-carbon-emission electricity to produce especially multicarbon (C 2+) products could contribute to a carbon-neutral to carbon-negative economy. In the O2 Lewis structure, there is a double bond between two oxygen atoms. They can be identified by using a Lewis structure. Lone pair of electrons are found in the outermost electron shell of atoms. The lone pair of electrons are unshared valence electrons. Oxygen also possesses a lone pair of electrons.  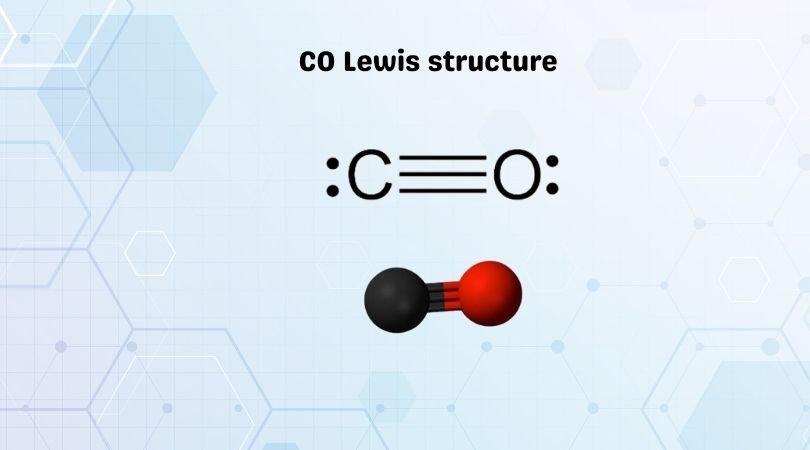
Similarly, to complete its octet, oxygen shares three valence electrons with carbon. By sharing three valence electrons, carbon forms three tipple bonds with oxygen. The overall carbon to oxygen atom ratio in a CO dot structure is 1:1. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
